In the past decade, only 3 HIV-2 positive cases have been reported by our laboratory, but none of them was a Singapore resident. Results are interpreted according to the manufacturer’s instructions and HIV-2 cases will be reported accordingly. Specifically, we use the HIV Blot 2.2 Western Blot assay (MP Biomedicals Asia Pacific, Singapore) which includes a HIV-2 specific peptide, reactivity to which will prompt follow-up testing using a HIV-2 confirmatory assay. We use the triple test algorithm, as recommended by WHO and similar to Australia, which also has a low HIV-2 prevalence, the western blot is employed as one of the confirmatory tests. Our laboratory confirms the status of all samples screened reactive for HIV in the country. The latest national statistic shows that in 2016, 103.7 per million resident population were diagnosed with HIV infection. Singapore has a low prevalence of HIV, with an exceedingly low prevalence of HIV-2. The revised algorithms will allow appropriate treatment according to the HIV type causing the infection. In the revised recommendations from the US and UK, a HIV-1/HIV-2 differentiation assay has been included to allow diagnosis of HIV-2 infection as the virus has an intrinsic resistance to non-nucleoside reverse transcriptase inhibitors as well as to some protease inhibitors to use tests that detect only HIV-1 may cause HIV-2 cases to be missed. As regards Australia, the Public Health Laboratory Network recommends that repeatedly reactive HIV antibody or antigen/antibody cases should be confirmed using a supplemental assay, inherently a western blot or a line probe immunoassay, with use of a nucleic acid or antigen test as necessary. On a similar note, HIV screening and confirmation guidelines in the United Kingdom (UK) published by Public Health England (PHE) also recommended 2 separate reactive fourth-generation HIV Ag/Ab screening tests for HIV screening, followed by a HIV-1/HIV-2 antibody differentiation immunoassay or a further immunoassay and then a HIV-1/HIV-2 typing assay for HIV confirmation. However, if the HIV prevalence in the population tested is less than 5%, a diagnosis of HIV-positive should be issued to individuals with 3 sequential reactive tests to enhance the positive predictive value. In 2015, the World Health Organization (WHO) reiterated its earlier recommendation that in settings where the HIV prevalence in the population tested is greater than 5%, a diagnosis of HIV-positive should be issued to individuals with 2 sequential reactive tests.  
The Centers for Disease Control and Prevention (CDC) in the United States (US) updated its recommendations for HIV confirmation in 2014, such that after an initial fourth-generation HIV Ag/Ab screening test, the use of HIV western blot was replaced by a HIV-1/HIV-2 antibody differentiation assay as the supplemental test for HIV confirmation. In recent years, revised algorithms for Human Immunodeficiency Virus (HIV) confirmatory testing have been recommended by various institutions worldwide. In that regard, of note is our observation of non-specific reactivity of lipaemic specimens to the HIV-2 gp140 band in the Geenius assay, which should prompt caution when interpreting results of such specimens. In conclusion, INNO and Geenius had comparable test performance, although the ease of use and shorter assay time for Geenius may make it the preferred choice for laboratories. Hence, further testing by a nucleic acid test or a p24 antigen test of specimens reactive on screening with a fourth generation Ag/Ab assay that are negative on confirmatory testing for HIV-specific antibody, may be useful. Nevertheless, overall poor sensitivity (25.5% - 44.7%) of these assays for the detection of early cases was observed, likely because these cases had very low or non-detectable levels of HIV antibodies. When early HIV cases were considered, INNO and Geenius were more likely to confirm an early-stage infection as positive. 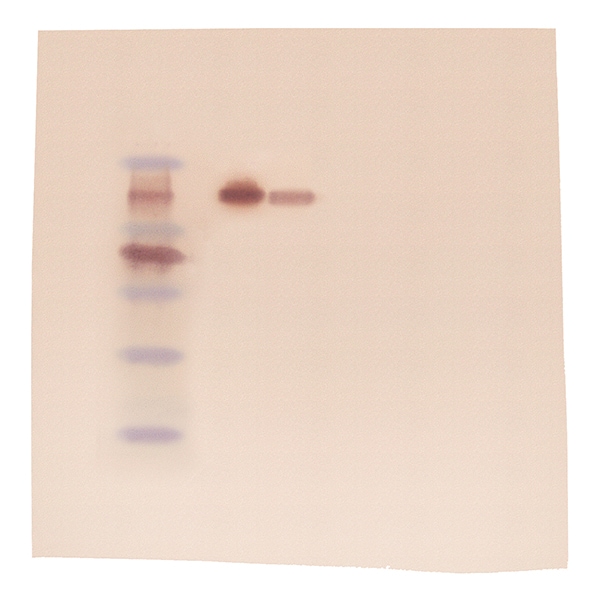
However, less non-specific reactivity was observed with the INNO and Geenius assays, where both of them were able to resolve MP-WB indeterminate cases. We show that all 3 assays had comparable test sensitivity in the detection of HIV-1 positive cases. Using the HIV Blot 2.2 (MP-WB), the INNO-LIA HIV I/II Score (INNO), and the Geenius HIV 1/2 Confirmatory Assay (Geenius), we tested 199 HIV-1 positive, 161 HIV negative, 65 HIV western blot indeterminate, 26 HIV seroconversion, 34 early HIV infection and 4 HIV-2 positive archived specimens. In view of recent revised recommendations for human immunodeficiency virus (HIV) confirmatory testing, the performance of 3 HIV confirmatory assays was compared. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
